 Multiple Choice Questions
Multiple Choice QuestionsAmong the following, the isoelectronic specie (s) is/are
(i) O2-, F-, Na+, Mg2+
(ii) Na+, Mg+, Al3+, F-
(iii) N3-, O2-, F-, Ne
(i) and (ii)
(i), (ii) and (iii)
(ii) and (iii)
(i) and (iii)
Match the following
| List I | List II |
| (a) PCl3 | (i) Square planar |
| (b) BF3 | (ii) T-shape |
| (c) ClF3 | (iii) Trigonal pyramidal |
| (d) XeF4 | (iv) See- saw |
| (v) Trigaonal planar |
(a) (b) (c) (d)
(iv) (ii) (i) (iii)
(a) (b) (c) (d)
(iii) (v) (ii) (iv)
(a) (b) (c) (d)
(iii) (v) (ii) (i)
(a) (b) (c) (d)
(iii) (v) (ii) (v)
If the kinetic energy in J, of CH4 (molar mass =16 g mol-1) at T(K) is X, the kinetic energy in J, of O2, (molar mass = 32 g mol-1) at the same temperature is
X
2X
X2
X/2
The given figure shows the Maxwell distribution of molecular speeds of a gas at three different temperatures T1,T2, and T3. The correct order of temperature is

T1 > T2 > T3
T1 > T3 > T2
T3 > T2 > T1
T2 > T3 > T1
The hybridisation of each carbon in the following compound respectively is CH3-CO-CH2CN
sp3,sp2,sp3,sp
sp3,sp3,sp2,sp
sp3,sp2,sp, sp3
sp2,sp, sp3, sp3
A.
sp3,sp2,sp3,sp
In the compound,
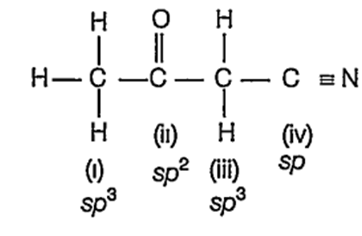
The C-atoms that is bonded to four different atoms involve four orbitals (1 of s and 3 of p- orbitals) during hybridisation. Hence, they have sp3 -hybridisation as in cases of (i) and (iii).
The C- atom bonded to three different atoms involves three orbitals (1 of s and 2 ofp-orbitals) Hence, the sp2-hybridisation in case of (ii).
The C-atom bonded to two different atoms involves two orbitals (1 of s and 1 of p-orbitals). Hence, the sp- hybridisation in case of (iv).
Given that N2(g) + 3H2(g) → 2NH3(g): ΔrH° =-92kJ, the standard molar enthalpy of formation in kJ mol-1 of NH3(g) is
-92
+46
+92
-46
Which one of the following is correct?
The equilibrium constant (Kc) is independent of temperature
The value of Kc is independent of initial concentrations of reactants and products
At equilibrium, the rate of the forward reaction is twice the rate of the backward reaction.
The equilibrium constant (Kc) for the reaction
