What are the necessary and sufficient conditions for a compound to exhibit geometrical isomerism ?
Conditions for geometrical isomerism: There are two necessary conditions for a compound to possess geometrical isomerism:
(i) It must contain a carbon-carbon double bond in the molecule.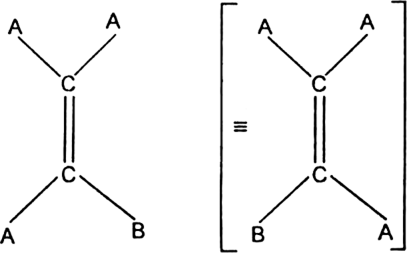
(ii) Two unlike atoms or groups must be linked to each doubly bonded carbon atoms.
Geometrical isomerism among alkenes does not occur when the doubly bonded carbon carry identical atoms or groups. For example, AAC = CAB does not exhibit geometrical isomerism.
