 Multiple Choice Questions
Multiple Choice QuestionsThe increasing order of acidity is :
CHCl2COOH , CH2ClCOOH , CH3COOH
CH3COOH , CHCl2COOH , CH2ClCOOH
CH2BrCOOH , CH2ClCOOH , CH3COOH
CH2BrCOOH , CH2ClCOOH , CHCl2COOH
Which of the following reaction will not produce ethylene glycol?
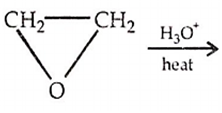
ClCH2CH2Cl
HO-CH2-CH2-OCH3
CH2 = CH2
Assertion : Mixture of benzaldehyde and acetaldehyde in hot allkaline medium gives cinnamaldehyde.
Reason : Benzaldehyde is strong electrophile than acetaldehyde.
If both assertion and reason are true and reason is the correct explanation of assertion
If both assertion and reason are true but reason is not the correct explanation of assertion.
If assertion is true but reason is false.
If both assertion and reason are false.
An aromatic compound C7H6Cl2 (A), gives AgCl on boiling with alcoholic AgNO2 solution and yields C,HOCI on treatment with sodium hydroxide. (A) on oxidation gives monochlorobenzoic acid. The compound (A) is:




Formic acid can be distinguished from acetic acid by its reaction with :
NaHCO3
Tollen's reagent
NaOH
None of these
Cannizaro reaction is not given by :
acetaldehyde
formaldehyde
benzaldehyde
triethyl acetaldehyde
B.
formaldehyde
Aldehydes containing no - H atom , on reaction with 50% NaOH or KOH , undergo disproportionation reaction to give an alcohol and sodium or potassium salt of acid .This eaction is called Cannizaro reaction . CH3CHO does not give Cannizaro reaction due to the presence of - H atoms.
In the Rosenmund's reaction
RCOCl + H2 RCHO + HCl
here BaSO4
promotes catalytic activity of Pd
removes the HCl formed in the reaction
deactivates Pd
activates Pd
