CBSE Multiple Choice Questions
Multiple Choice QuestionsWhich of the following is least likely to behave as Lewis base?
NH3
BF3
OH-
H2O
Which of the following structures is the most preferred and hence of lowest energy of SO3 ?




In which of the following molecules the central atom does not have sp3 hybridisation?
CH4
SF4
BF4-
BF4-
In which one of the following species the central atom has the type of hybridisation which si not the same as that present in the other three?
SF4
I3-
SbCl52-
SbCl52-
The correct order of the decreasing ionic radii among the following isoelectronic species is
Ca2+ > K+ >S2- > Cl-
Cl- > S2- > Ca2+ > K+
S2- > Cl- > K+ > Ca2+
S2- > Cl- > K+ > Ca2+
In which of the following pairs of molecules/ions, the central atoms have sp2 hybridization?
NO2- and NH3
BF3 and NO2-
NH2- and H2O
NH2- and H2O
Which one of the following species does not exist under normal conditions?
Be2+
Be2
B2
B2
Decreasing order of stability of  is
is
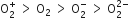
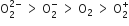
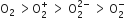
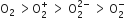
Which of the following has the minimum bond length?
O2-
O22-
O2
O2
In which of the following pairs, both the species are not isostructural?
SiCl4.PCl4+
Diamond, carbide
NH3, PH3
NH3, PH3

