CBSE Multiple Choice Questions
Multiple Choice QuestionsThe function of AlCl3, in Friedel-Craft's reaction is to
absorb HCl
absorb water
produce nucleophile
produce electrophile.

What is B ?
![]()
![]()
![]()

Decreasing order of stability of ions is.
(i) CH3 - H - CH3
(ii) CH3 - H - OCH3
(iii) CH3 - H - COCH3
(i) > (ii) > (iii)
(ii) > (i) > (iii)
(ii) > (iii) > (i)
(iii) > (i) > (ii)
In which of the following preparations of ether, the configuration about chiral centre is not retained?
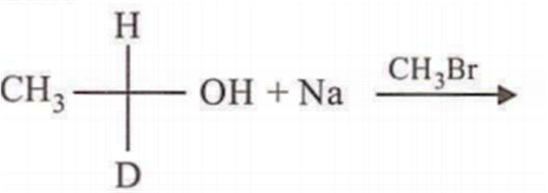


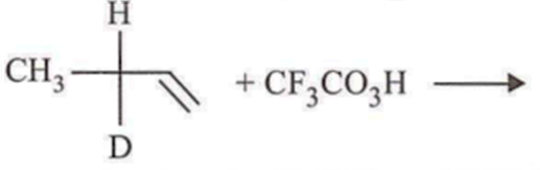
Which ofthe following compounds is not chiral?
1-Chloropentane
2-Chloropentane
1-Chloro-2-methyl pentane
3-Chloro-2-methyl pentane
Which ofthe following molecules will not show optical activity?
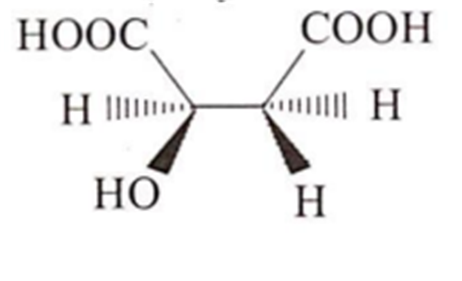

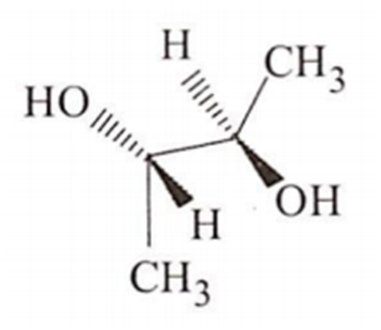

Williamson's synthesis involves :
SN1 mechanism
nucleophilic addition
SN2 mechanism
SE mechanism
In one reaction,![]() is treated with CH3CN and then hydrolysed. In another reaction
is treated with CH3CN and then hydrolysed. In another reaction ![]() is treated with ethyl acetate and then hydrolysed.
is treated with ethyl acetate and then hydrolysed.

A and B are
![]()
![]()
![]()

Which of these compounds is synthesised by chloral?
DDT
BHC
Chloroform
Michlers ketone.
Consider the reaction,

The correct explanation is
the product is formed due to nucleophilic substitution
the product is formed according to Saytzeff's rule
conjugated double bond product is formed, due to higher stability because of resonance stabilization
(CH3)3CO- is a better leaving group

