CBSE Multiple Choice Questions
Multiple Choice QuestionsOn addition of conc. H2SO4 to a chloride salt, colourless fumes are evolved but in case of iodide salt, violet fumes come out. This is because
H2SO4 reduces HI to I2
HI is of violet colour
HI gets oxidised to I2
HI changes to HIO3
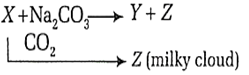
The chemical formulae of X, Y and Z are
| X | Y | Z |
| CaO | Ca(OH)2 | NaOH |
| NaOH | CaO | CaCO3 |
| NaOH | Ca(OH)2 | CaCO3 |
| Ca(OH)2 | NaOH | CaCO3 |
What is correct about the following structure?
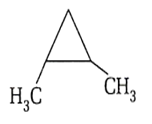
Total stereoisomers = 4
Number of chiral carbons = 1
Number of optical isomers= 2
Number of meso compounds = 2
During estimation ofnitrogen in the organic compound by Kjeldahl's method, the ammonia evolved from 0.5 g of the compound in Kjeldahl's estimation of nitrogen, neutralised 10 mL of 1M H2SO4. Find out the percentage of nitrogen in the compound.
14%
28%
56%
68%
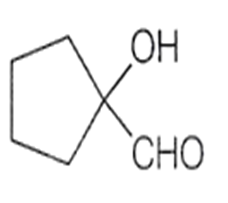
What is product of the following sequence of reactions?




Which one of the following is aromatic?
Cyclopentadienyl cation
Cyclooctatetraene
Cycloheptatriene
Cycloheptatrienylcation
Acetic anhydride is prepared in the laboratory by heating sodium acetate with
ethyl chloride
acetyl chloride
conc. H2SO4
zinc dust
Affinity for hydrogen decreases in the group from fluorine to iodine. Which ofthe halogen acids should have highest bond dissociation enthalpy?
HF
HCl
HBr
HI
Which of the following compounds is soluble in benzene but almost insoluble in water?
C2H5OH
CH3CO2H
CH3CHO
C6H5NO2
The complexes [Co(NH3)6][Cr(CN)6] and [Cr(NH3)6][Co(CN)6] are the examples of which type of isomerism?
Ionisation isomerism
Coordination isomerism
Geometrical isomerism
Linkage isomerism

