 Short Answer Type
Short Answer TypeGive reactions and the conditions required for preparation of the following compounds:
(i) XeF6
(ii) XeOF4
Carry out the following conversions:
(i) Methyl chloride to acetic acid.
(ii) Benzene to benzoic acid
(iii) Ethanol to acetone.
Deficiency of what vitamins will cause the following diseases:
(i) Night blindness.
(ii) Scurvy
Give balanced equations for the following:
(i) Glycerol is heated with oxalic acid at 110° C (383K).
(ii) Acetamide is heated with sodium hydroxide.
(iii) Acetone reacts with hydrogen in the presence of heated copper.
Give one good chemical test to distinguish between the following pairs of organic compounds:
(i) Benzaldehyde and acetone.
(ii) Methylamine and dimethylamine.
Give an example (equation) for each of the following name reactions:
(i) Aldol condensation.
(ii) Reimer-Tiemann reaction.
(iii) Rosenmund’s reductions.
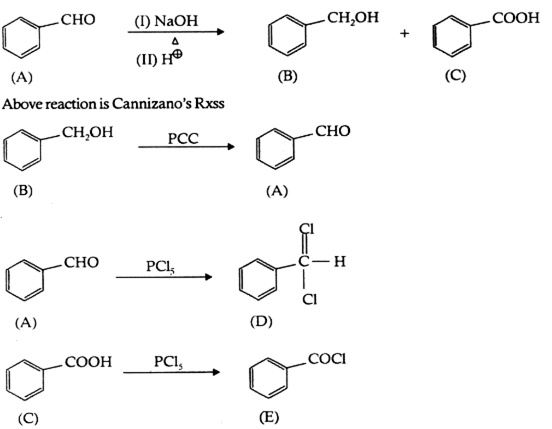
An organic compound A has the molecular formula of C7H6O. When A is treated with NaOH followed by acid hydrolysis, it gives two products, B and C. When B is oxidised, it given A, When A and C are each treated separately with PC15, they give two different organic products D and E.
(i) Identify A to E.
(ii) Give the chemical reaction A is treated with NaOH and name the reaction.
A’ can be an aldehyde with the absence of α-Hydrogen because ‘A’ gives Cannizzaro Rxn. Hence
C7H6O = C6H5CHO
Draw a pair of isomers for each of the following and name the type of isomerism:
(i) C4H10
(ii) C2H2C12
(iii) CH3COCH3
(iv) C4H10O
