 Short Answer Type
Short Answer TypeState Bohr’s postulate to define stable orbits in hydrogen atom. How does de Broglie’s hypothesis explain the stability of these orbits?
Bohr's postulate:
An atom has a number of stable orbits in which an electron can reside without the emission of radiant energy. Each orbit corresponds, to a certain energy level.
Electrons revolve in a circular orbit. Centripetal force is provided by electrostatic force between electron and proton.
Given as,
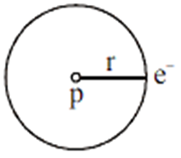
The motion of an electron in a circular orbit is restricted in such a manner that its angular momentum is an integral multiple of h/2π, Thus
3.
An electron may jump spontaneously from one orbit (energy level E1) to the other orbit (energy level E2) (E2 > E1); then the energy change AE in the electron jump is given by Planck’s equation
∆E = E2-E1 = hv
Where h = Planck’s constant.
And v = frequency of light emitted.
When the electron jumps from nth higher orbit to pth lower orbit it emits energy in form of the photon.
En - Ep = hv
According to de'Broglie hypothesis
A hydrogen atom initially in the ground state absorbs a photon which excites it to the n = 4 level. Estimate the frequency of the photon.
Explain the processes of nuclear fission and nuclear fusion by using the plot of binding energy per nucleon (BE/A) versus the mass number A.
A radioactive isotope has a half-life of 10 years. How long will it take for the activity to reduce to 3.125 %?
A student wants to use two p-n junction diodes to convert alternating current into direct current. Draw the labelled circuit diagram she would use and explain how it works.
Draw the typical input and output characteristics of an n-p-n transistor in CE configuration. Show how these characteristics can be used to determine (a) the input resistance (ri), and (b) current amplification factor.
Give three reasons why the modulation of a message signal is necessary for long-distance transmission.
Draw a ray diagram to show image formation when the concave mirror produces a real, inverted and magnified image of the object.
