 Short Answer Type
Short Answer TypeHow will you convert the following (write the relevant equations) :
Glucose to osazone.
 Long Answer Type
Long Answer TypeOsmotic Pressure. The pressure which is applied to the solution in order to prevent the passage of solvent into it through a semi-permeable membrane present between the two is termed as osmotic pressure.
We know that osmotic pressure is a colligative property and hence it depends on the number of solute particles.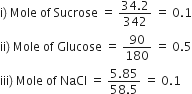
Since 1 mole = 6.023 x 1023 particles
So Sucrose = 6.023 x 1022 particles
(As Sucrose and Glucose are undissociated)
Glucose = 30.115 x 1022 particles
NaCl = 12.046 x 1022 particles.
(NaCl dissociates to give two ions)
Hence, the increasing order of their osmotic pressure is, Sucrose < NaCl < Glucose.
A cell is constructed by dipping a zinc rod in 0.1 M zinc nitrate solution and a lead rod in 0.2 M lead nitrate solution. E°pb2+/pb =-0.13 V and E°Zn2+/ Zn=–0.76 V. [3]
(i) Write the spontaneous cell reaction.
(ii) Calculate standard emf and emf of the cell.
(b) Explain the purification of common salt by bubbling hydrogen chloride gas through its solution.
Give one test each to distinguish between the following pairs. Write the relevant equations.
(i) Benzoic acid and phenol
(ii) Glucose and fructose
(iii) Acetophenone and benzophenone.
