 Short Answer Type
Short Answer TypeHow will you convert the following (write the relevant equations) :
Glucose to osazone.
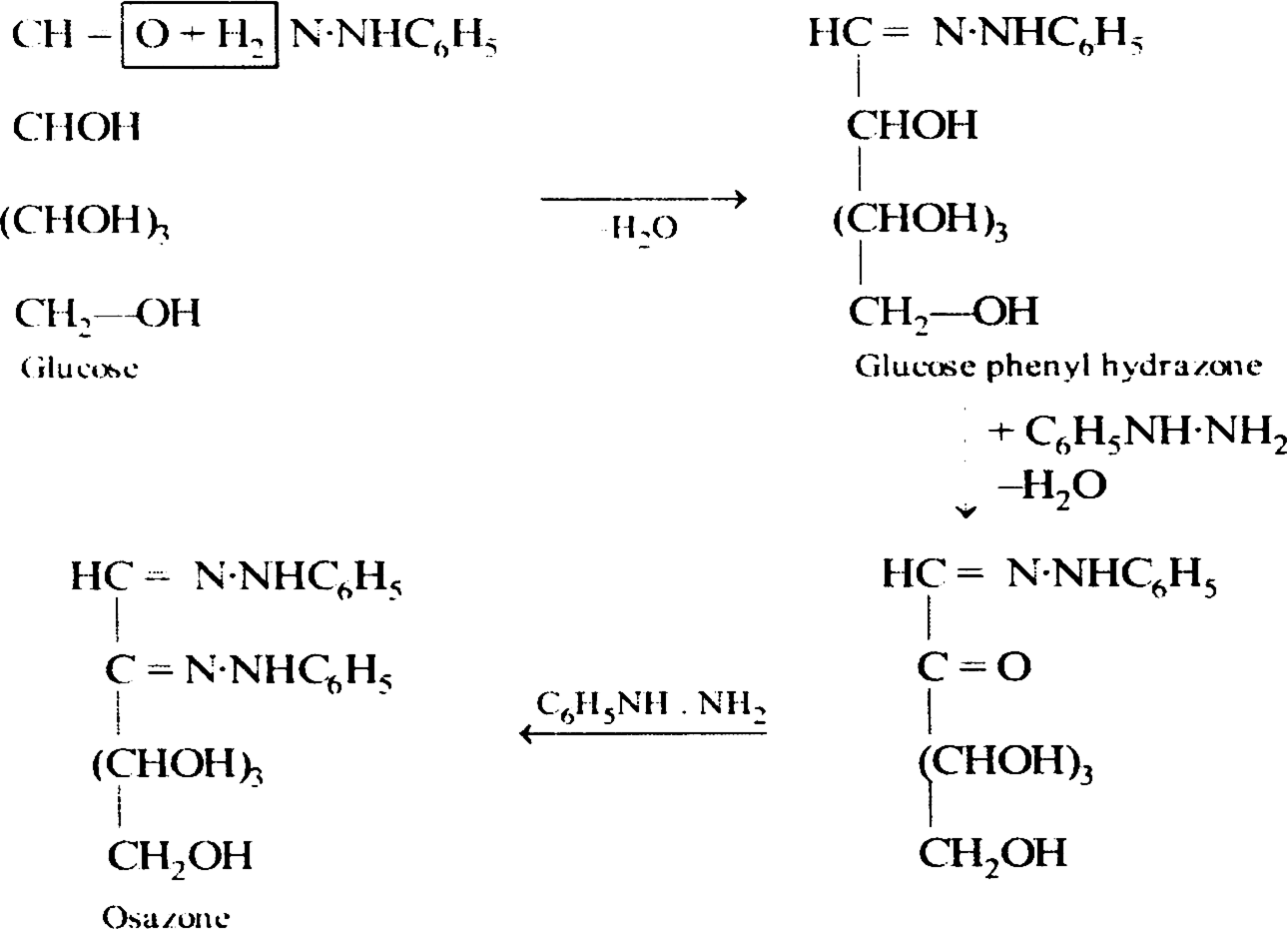
 Long Answer Type
Long Answer TypeA cell is constructed by dipping a zinc rod in 0.1 M zinc nitrate solution and a lead rod in 0.2 M lead nitrate solution. E°pb2+/pb =-0.13 V and E°Zn2+/ Zn=–0.76 V. [3]
(i) Write the spontaneous cell reaction.
(ii) Calculate standard emf and emf of the cell.
(b) Explain the purification of common salt by bubbling hydrogen chloride gas through its solution.
Give one test each to distinguish between the following pairs. Write the relevant equations.
(i) Benzoic acid and phenol
(ii) Glucose and fructose
(iii) Acetophenone and benzophenone.