 Short Answer Type
Short Answer TypeActivation energy. Excess of energy which must be supplied to reactant to undergo the chemical reaction called activation energy.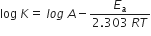
where, K = rate constant
A = Arrhenius constant
Ea = Activation energy
R = Gas constant
T = Temperature (K)
The slope of the line is equal to 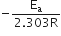

(i) 10 g of an organic compound was present in 100 cm3 of water. It was extracted twice with fresh 50 cm3portions of ether. How much organic compound was extracted ? (The organic compound is three times more soluble in ether than in water.)
(ii) Give one example each of homogeneous and heterogeneous catalysis.
(i) Name the crystal structure of the copper metal.
(ii) What is the coordination number of copper in its crystalline state ?
ΔH and ΔS for vaporization of water at 1 atm pressure are 40.63 KJ mole 1 and 108.8 JK– 1mole– 1 respectively. Calculate the temperature at which the free energy change for the transformation will be zero. Predict the sign of free energy change below this temperature.
(ii) The heat supplied to a Carnot engine is 1958.22 KJ. How much useful work can be done by the engine which works between 0 °C and 100 °C ?
Write balanced chemical equations for each of the following reactions :
(i) Sodium thiosulphate with aqueous silver nitrate.
(ii) Copper sulphate with aqueous potassium iodide.
(iii) Hydrogen peroxide with acidified ferrous sulphate solution.
(iv) Ozone with moist iodine.
Give reasons :
(i) Hydrogen peroxide is used for restoring the colour of lead paintings.
(ii) Silicones are used as lubricants at both high and low temperatures.
