 Short Answer Type
Short Answer TypeGive a reason for each of the following :
The number of hydrogen ions in an aqueous solution of acetic acid increase considerably with dilution while this is not the case with an aqueous solution of hydrogen chloride.
Give a reason for each of the following :
A mixture of NH4Cl and NH4OH is used to precipitate the metallic hydroxides of group III.
The solubility product of hydroxides of group III are low as compared to those of group IV, V and VI. So, it is must to have only those many OH- ions which are necessary to precipitate group III hydroxides only. NH4C1 is a strong electrolyte and NH4OH is a weak electrolyte. When NH4C1 is added before NH4OH, it suppresses the dissociation of NH4OH due to common ion effect and decreases the concentration of OH- ions.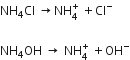
Therefore, a mixture of NH4C1 and NH4OH is used to precipitate the metallic hydroxides of group III.
Arrange the following in increasing order of acidity and explain your order:
Phenol, Methanol, Water.
