 Short Answer Type
Short Answer TypeWhy are neutrons considered as ideal particles for nuclear fission reactions ? Calculate the energy released in the following nuclear reaction.
1H2 + 1H2 →2H4 +energy
(mass of 1H2 = 2.014102 u, mass of 2He4 = 4.002604 u).
Neutrons are used as projectiles in nuclear reaction, which initiates nuclear reactions and are present in product to carry the reaction further.
It is considered as ideal particles for nuclear reaction, because they are neutral particles on which neither any force of attraction nor any force of repulsion acts.
Mass of reactants = 2 x 2.014102 U = 4.028204 U
Mass of product = 4.002604 V
Mass defect = 4.028204 - 4.002604
Δm = 0.0256 U
Energy released = 0.0256 x 931 MeV
= 23.8336 MeV
 Long Answer Type
Long Answer Type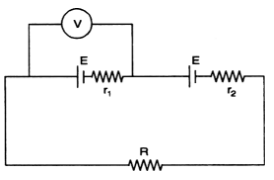
(i) With proper labelling of the axis, draw the input and output characteristic curves of a npn transistor in common emitter configuration.
(ii) Define current gain for a transistor in common emitter configuration.
