 Short Answer Type
Short Answer TypeName a phenomenon or an experiment which proves.
(i) Particle nature of electro-magnetic radiations.
(ii) Wave nature of particles.
(i) State the postulate of Bohr's theory regarding:
(1) Angular momentum of an electron.
(2) Emission of a photon.
(ii) Total energy of an electron orbiting around the nucleus of an atom is always negative. What is the significance of this?
i) Bohr’s theory regarded that:
(1) Angular momentum of electron : Electrons can revolve only in those orbits in which their angular momentum is integral multiple of  .
.
Using the formula,
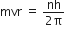
(2) Emission of Photon : When an electron receives energy from outside then one of its outer electron goes to same higher orbit while returning back the electron emit energy in form of photon.
(ii) Electrons is bound to nuclear energy must to supplied to it for ejection of electrons.
(i) Draw a labelled graph showing variation of relative intensity of X-rays versus their wavelength λ . Mark on the graph.
(ii) State how the value of can be varied.
(i) What is the significance of binding energy per nucleon of nucleus?
(ii) In a certain star, three alpha particles undergo fusion in a single reaction to form 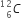 nucleus. Calculate the energy released in the reaction in MeV.
nucleus. Calculate the energy released in the reaction in MeV.
Show by drawing labelled diagrams, the nature of output voltages in case of:
(i) A half wave rectifier.
(ii) A full wave rectifier.
(iii) An Amplifier.
(In Each case, input in an ac voltage.)
Circuit diagrams of these devices are not required.
