 Multiple Choice Questions
Multiple Choice QuestionsCalculate H in kJ for the following reaction:
C (g) + O2 (g) → CO2 (g)
Given that,
H2O (g) + C (g) → CO (g) + H2 (g) ; H = +131 kJ
CO (g) + O2 (g) → CO2 (g); H = -282 kJ
H2 (g) + O2 (g) → H2O (g); H = -242 kJ
-393
+393
+655
-655
Boron halides behave as Lewis acids because of their .......... nature.
proton donor
covalent
electron deficient
ionising
The correct order of reducing abilities of hydrides of V group elements is
NH3 < PH3 < AsH3 < SbH3 < BiH3
NH3 > PH3 > AsH3 > SbH3 > BiH3
NH3 < PH3 > AsH3 > SbH3 > BiH3
SbH3 > BiH3 > AsH3 > NH3 > PH3
The number of sigma and pi bonds in peroxodisulphuric acid are, respectively
9 and 4
11 and 4
4 and 8
4 and 9
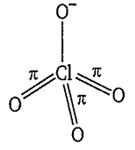
The compound in which the number of d-p bonds are equal to those present in ClO
XeF4
XeO3
XeO4
XeF6
B.
XeO3
The structure of ClO consists of 3 d-p bonds.
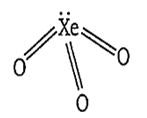
XeO3 also consists of 3 d-p bonds and its structure is as follows-
Which one of the following reactions represents the oxidising property of H2O2?
2KMnO4 + 3H2SO4 + 5H2O2 → K2SO4 + 2MnSO4 + 8H2O + 5O2
2K3[Fe(CN)6] + 2KOH + H2O2 → 2K4[Fe(CN)6] + 2H2O + O2
PbO2 + H2O2 → PbO + H2O + O2
2KI + H2SO4 + H2O2 → K2SO4 + I2 + 2H2O
Which of the following statements are correct for alkali metal compounds ?
(i) Superoxides are paramagnetic in nature.
(ii) The basic strengths of hydroxides increases down the group.
(iii) The conductivity of chlorides in their aqueous solutions decreases down the group.
(iv) The basic nature of carbonates in aqueous solutions is due to cationic hydrolysis.
(i), (ii) and (iii) only
(i) and (ii) only
(ii), (iii) and (iv) only
(iii) and (iv) only
[Co(NH3)5SO4]Br and [Co(NH3)5Br]SO4 are a pair of ....... isomers.
ionisation
ligand
coordination
hydrate
Among the following compounds, which one is not responsible for depletion of ozone layer?
CH4
CFCl3
NO
Cl2