 Multiple Choice Questions
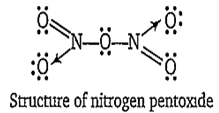
Multiple Choice QuestionsThe bond present in N2O5 are
only ionic
covalent and coordinate
only covalent
covalent and ionic
B.
covalent and coordinate
Nitrogen pentoxide N2O5) has both covalent as well as coordinate bonds. In the gaseous state, it exists as a symmetrical molecule having the structure O2N-O-NO2. The N-O-N bond is almost linear. X-ray studies reveals the ionic nature ofsolid N2O5 i.e., nitronium nitrate, NaNO.
5 millimoles of caustic potash and 5 millimoles of oxalic acid are mixed and dissolved in 100 mL water. The solution will be
basic
acidic
neutral
cannot say
How many moles of O2 can be obtained by electrolysis of 90 g H2O?
5.0 mol
0.5 mol
2.5 mol
0.25 mol
Four different sets of quantum numbers for 4 electrons are given below:
e1 = 4, 0, 0, -
e2 = 3, 1, 1, -
e3 = 3, 2, 2, +
e4 = 3, 0, 0, +
The order of energy of e1, e2, e3, e4 is
e1 > e2 > e3 > e4
e4 > e3 > e2 > e1
e3 > e1 > e2 > e4
e2 > e3 > e4 > e1
Electron affinity is positive when
O- is formed from O
O2- is formed from O-
O+ is formed from O
electron affinite is always a negative value
When solid melts, there will be
a decrease in enthalphy
a decrease in free energy
a decrease in entropy
all the above factors remain constant
Which solution is a buffer?
Acetic acid + NaOH (equimolar ratio)
Acetic acid + NaOH (1 : 2 molar ratio)
Acetic acid + NaOH (2 : 1 molar ratio)
HCl + NaOH (equimolar ratio)
