 Multiple Choice Questions
Multiple Choice QuestionsSpectrum of sunlight is an example for
continuous absorption spectrum
band emission spectrum
line absorption spectrum
continuous emission spectrum
In hydrogen atom, electron excites from ground state to higher energy state and its orbital velocityis reduced to rd of its initial value. The radius of the orbit in the ground state is R. The radius of the orbit in that higher energy
9 R
2 R
3 R
27 R
Decay constants of two radio-active samples A and B are 15x and 3x respectively. They have equal number of initial nuclei. The ratio of the number of nuclei left in A and B after a time is
e-2
e
e2
e-1
Mass number of the elements A, B, C and D are 30, 60, 90 and 120 respectively. The specific binding energy of them are 5 MeV, 8.5 MeV, 8 MeV and 7 MeV respectively. Then, in which of the following reaction/s energy is released ?
1. D → 2B
2. C → B + A
3. B → 2A
in (1), (2) and (3)
Only in (1)
in (2), (3)
in (1), (3)
Copper and Germanium are cooled from room temperature to 100 K. Then, the resistance of
germanium increases, copper increases
germanium decreases, copper increases
germanium decreases, copper decreases
germanium increases, copper decreases
Frequencies of light incident on a system of scattering particles are in the ratio of 1 : 2. Then, the intensity of scattered light in a particular directions is
1 : 16
1 : 4
1 : 2
1 : 8
A.
1 : 16
The ratio of the magnetic dipole moment to the angular momentum of the electron in the 1st orbit of hydrogen atom is
White light is incident normally on a glass slab. Inside the glass slab
all colours travel with the same speed
red light travels faster than other colours
violet light travels faster than other colours
yellow light travels faster than other colours
Two thin plano-convex lenses each of focal length f are placed as shown in the figure. The ratio of their effective focal lengths in the three cases is
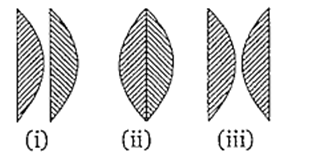
3 : 2 : 1
1 : 2 : 3
1 : 2 : 1
1 : 1 : 1
