 Multiple Choice Questions
Multiple Choice QuestionsAssertion : In a mixture of Cd(II) and Cu(II),Cd2+ gets precipitated in presence of KCN by H2S.
Reason : The stability constant of [Cu(CN)4]3- is greater than [Cd(CN)4]2-.
If both assertion and reason are true and reason is the correct explanation of assertion.
If both assertion and reason are true but reason is not the correct explanation of assertion.
If assertion is true but reason is false.
If both assertion and reason are false.
Assertion : The presence of a large number of Schottky defects in NaCl lowers its density.
Reason : In NaCl, there are approximately 106 Schottky pairs per cm3 at room temperature.
If both assertion and reason are true and reason is the correct explanation of assertion.
If both assertion and reason are true but reason is not the correct explanation of assertion
If assertion is true but reason is false.
If both assertion and reason are false.
Assertion : Magnesium is extracted by the electrolysis of fused mixture of MgCl2, NaCl and CaCl2.
Reason : Calcium chloride acts as a reducing agent.
If both assertion and reason are true and reason is the correct explanation of assertion.
If both assertion and reason are true but reason is not the correct explanation of assertion.
If assertion is true but reason is false.
If both assertion and reason are false.
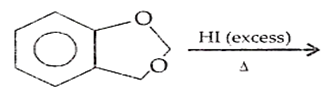
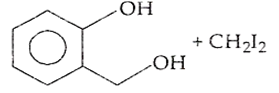
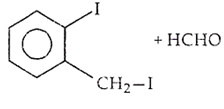
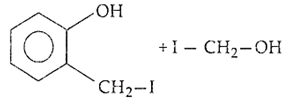
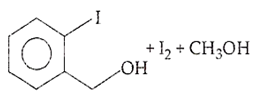
B.

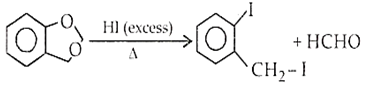
Ethers are readily cleaved by action of HI to form alcohol and alkyl halide.
R - O - R + HX RX + R - OH
If excess of halogen acid is used, then alcohol formed reacts further with halogen acid to produce alkyl halide.
Which of the following reaction will not produce ethylene glycol?
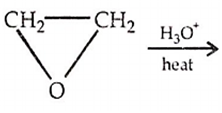
ClCH2CH2Cl
HO-CH2-CH2-OCH3
CH2 = CH2
Salicylic acid can be easily prepared by reaction between
phenol and CO2
benzoic acid and H2O2
benzene diazonium chloride and CO2
phenol and formic acid.
Reaction of aniline with HNO2 followed by treatment of dilute acid gives
C6H5NHOH
C6H5OH
C6H5NHNH2
C6H6
