 Multiple Choice Questions
Multiple Choice QuestionsPressure versus temperature graph of an ideal gas is as shown in figure. Density of the gas at point A is ρ0 Density at point B will be
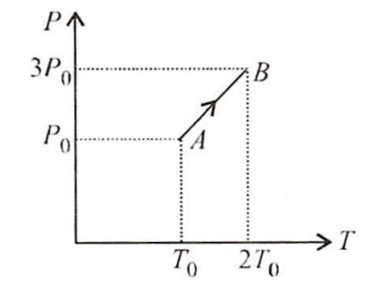
ρ0
ρ0
ρ0
2ρ0
The latent heat of vaporisation of a substance is always
greater than its latent heat of fusion
greater than its latent heat of sublimation
equal to its latent heat of sublimation
less than its latent heat of fusion
A reversible engine converts one-sixth of the heat input into work. When the temperature of the sink is reduced by 62°C, the efficiency of the engine is doubled. The temperatures of the source and sink are
99oC, 37oC
80oC, 37oC
95oC, 37oC
90oC, 37oC
A.
99oC, 37oC
Maximum efficiency of an engine working between temperatures T2 and T1 is given by the fraction of the heat absorbed by an engine which can be converted into work is known as efficiency of the heat engine.
Mathematically
Efficiency η =
η =
⇒ 6 T2 6 T1 = T2
∴ T2 = 1.2 T1 .....(i)
Where T2 is the source temperature and T1 is the sink temperature
If the sink temperature ( T1 ) is reduced by 62oC, the efficiency gets doubled i.e
η = T2
= 2 ×
η =
6 T2 6 T1 + 372 = 2 T2
⇒ 6 T2 2 T2 6 T1 + 372 = 0
⇒ 4 T2 6 T1 + 372 = 0
Substituting the value of T2 from equatin (i), we will get
4 ( 1.2 T1 ) 6 T1 + 372 = 0
4.8 T1 6 T1 + 372 = 0
⇒ 372 = 1.2 T1
⇒ T1 = 310 K
Therefore,
T2 = 1.2 × 310
T2 = 372 k
Hence, the temperature of the source ane the sink is 372 K i.e 99oC and 310 K i.e 37oC respectively.
A liquid is kept in a cylindrical vessel which is being rotated about a vertical axis through the centre of the circular base. If the radius of the vessel is r and angular velocity of rotation is w, then the difference in the heights of the liquid at the centre of the vessel and the edge is
A capillary tube of radius r is immersed in water and water rises in it to a height h. The mass of water in the capillary tube is 5 g. Another capillary tube of radius 2 r is immersed in water. The mass of water that will rise in this tube is
2.5 g
5.0 g
10 g
20 g
Five sinusoidal waves have the same frequency 500 Hz but their amplitudes are in the ratio 2 : and their phase angles 0, respectively. The phase angle of resultant wave obtained by the superposition of these five waves is
30o
45o
60o
90o
The second overtone of an open pipe has the same frequency as the first overtone of a closed pipe 2 m long. The length of the open pipe is
8 m
4 m
2 m
1 m
Let T1 and T2 be the time periods of springs A and B when mass M is suspended from one ·end of each spring. If both springs are taken in series and the same mass M is suspended from the series combination, the time period is T then
T1 + T2 +T3
T2 =
Assertion: Water kept in an open vessel will quickly evaporate on the surface of the moon.
Reason: The temperature at the surface of the moon is much higher than boiling point of water.
If both assertion and reason are true and reason is the correct explanation of assertion.
If both assertion and reason are true but reason is not the correct explanation of assertion.
If assertion is true but reason is false.
If both assertion and reason are false.
