How are particles of matter affected with increasing or reducing pressure on the matter at a given temperature ?
On increasing pressure, particles of a matter come closer and these move apart on reducing pressure at a given temperature. Thus if pressure is increasingly applied on a gas, particles of the gas come closer and closer and eventually the gas may change into liquid and then solid form. Fig. shows three cylinders in which a matter is present as gas, liquid and solid.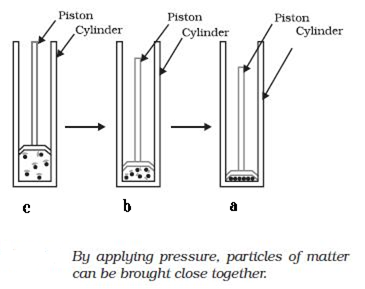
Changing state of matter on application of pressure : (a) solid; (b) liquid; (c) gas.
