Physical characteristics of acid and base are:
Acids are
(i) sour in taste.
(ii) change the colour of blue litmus to red.
Bases are
(i) bitter in taste.
(ii) change the colour of red litmus to blue.
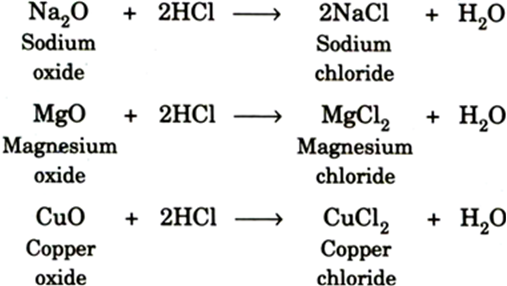
Chemical Reactions:
Marble is calcium carbonate. It reacts with HCl to give CO2
CaCO3 + 2HCl CaCl2 + CO2 + H2O
Marble (A)
CO2 when passed through lime water [Ca(OH)2] gives a white precipitate of CaCO3.
Ca(OH)2 + CO2(g) CaCO3(s) + H2O
White ppt.
(B)
On passing excess CO2, CaCO3 dissolves forming soluble Ca(HCO3)2.
CaCO3 + CO2 Ca(HCO3)2 (aq)
Calcium
hydrogen
carbonate
(C)
A — Carbon dioxide
B — Calcium carbonate
C — Calcium hydrogen carbonate
Complete and balance the following equations:
(i) H2CO3 + NaOH →
(ii) CH3COOH + NH4OH →
(iii) HNO3 + KOH →
(iv) H2SO4 + NaOH →
(a) What are these reactions called?
(b) Name the salt formed in each case.

(a) All the above are Neutralization Reactions.
Name of salts in reactions:
(b) (i) Sodium carbonate, (ii) Ammonium acetate,
(iii) Potassium nitrate, (iv) Sodium sulphate.