An organic compound A with molecular formula C7H8 on oxidation by chromylchloride in the presence of CCl4 gives a compound B which gives positive tollen’s test. The compound B on treatment with NaOH followed by acid hydrolysis gives two products C and D. C on oxidation gives B which on further oxidation gives D. The compound D on distillation with soda lime gives a hydrocarbon E. Below 60oC, concentrated nitric acid reacts with E in the presence of concentrated sulphuric acid forming a compound F. Identify the compounds A, B, C, D, E and F.
Identify the compounds from (A) to (F):
A = Toluene (or) C6H5CH3
B = Benzaldehyde (or) C6H5CHO
C = Benzyl Alcohol (or) C6H5CH2OH
D = Benzoic Acid (or) C6H5COOH
E = Benzene (or) C6H6
F = Nitrobenzene (or) C6H5NO2
An organic compound A has the molecular formula of C7H6O. When A is treated with NaOH followed by acid hydrolysis, it gives two products, B and C. When B is oxidised, it given A, When A and C are each treated separately with PC15, they give two different organic products D and E.
(i) Identify A to E.
(ii) Give the chemical reaction A is treated with NaOH and name the reaction.
A’ can be an aldehyde with the absence of α-Hydrogen because ‘A’ gives Cannizzaro Rxn. Hence
C7H6O = C6H5CHO
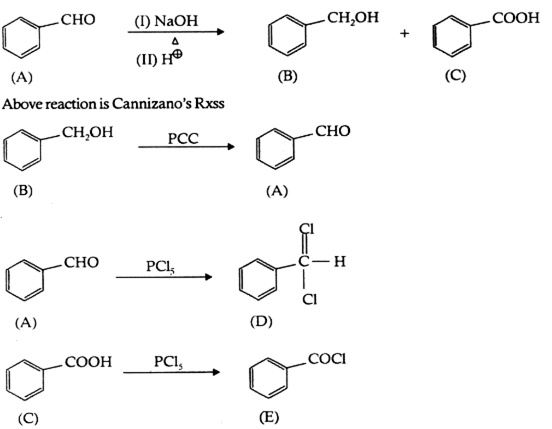
Give balanced equations for the following reactions:
(i) Benzaldehyde treated with hydrogen cyanide.
(ii) Chlorine is passed through diethyl ether.
(iii) Benzoic acid solution is treated with sodium carbonate.

Give one good chemical test to distinguish between the following pairs of organic compounds:
(i) Benzaldehyde and acetone.
(ii) Methylamine and dimethylamine.
i) Benzaldehyde gives the tollen test but acetone does not give this test.
(ii) Carbylamine test will be given by 1° Amine i.e. methylamine, not by dimethyl amine.
