 Short Answer Type
Short Answer TypeWe know that,
1 amu = 1.660565 x 10-27kg
Energy equivalent to this mass, E = mc2
E = (1.660565 x 10-27) x (2.998 x 108)2
E = 1.4925 x 10-10J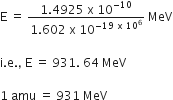
 Long Answer Type
Long Answer TypeFor Bohr’s model of the hydrogen atom, the energy of electron in its ground state is found to be-13.6 eV.
(i) Draw an energy level diagram for the hydrogen atom and mark the values of energy (in eV) at n = 2 and n = ∝.
(ii) Obtain the maximum energy of a photon emitted by the hydrogen atom in eV.
(i) On what does the minimum wavelength of the continuous x-ray spectrum depend
(ii) When the filament current is increased, the intensity of the x-ray produced increases. Why?
(iii) Why is the structure of crystals studied by x-rays ?
